Energy Systems: The Complete Science of Engineering Your Mitochondrial Power Plants for Limitless Cellular Energy and Longevity
Caffeine masks fatigue. It doesn’t produce energy. Every watt of cognitive and physical output runs on ATP — and your mitochondria produce it through three metabolic stages, each with different bottlenecks.
The ATP cycle mapped alongside mapped below shows why most people’s energy ceiling is set at stage three.
Three stages convert fuel into cellular energy — the bottleneck is usually stage three.
Your energy systems are the microscopic power plants inside every cell that convert food into usable fuel. Called mitochondria, these structures run a three-stage process to produce ATP — the molecule your body uses for everything from thinking to moving. When they’re healthy and abundant, energy feels limitless. When they degrade, fatigue sets in long before your muscles or mind should be tired.
The ATP Engine
Glycolysis, Krebs, and the ETC produce 38 ATP per glucose molecule — but 89% comes from the final stage. Understanding the pipeline is step one.
Mitophagy — Cellular Cleanup
Damaged mitochondria leak reactive oxygen species that destroy their neighbours. Mitophagy is the quality control system that clears the wreckage.
Substrate Flexibility
Flexible mitochondria switch between glucose and fatty acids on demand. Rigid ones stay locked on glucose — creating the crashes caffeine masks.
Hormesis — Stress Adaptation
Cold, heat, and high-intensity exercise trigger mitochondrial biogenesis via PGC-1α. The right dose of stress forces mitochondria to multiply.
Light & Circadian Signalling
Mitochondria have circadian rhythms tied to light. Morning sunlight primes cytochrome c oxidase in the ETC for peak daytime output.
TLDR: 10 Mitochondrial Tactics. 10 Energy Myths Busted.
Everything below distilled into 20 cards. Deploy the tactics, debunk the myths. The full science follows after.
10 Myths About Mitochondria
“Energy drinks and caffeine solve fatigue.”
Stimulants artificially block adenosine receptors (masking fatigue) but do not increase actual ATP production. Chronic use can worsen function by increasing oxidative stress and disrupting sleep.
“Mitochondrial health is just about diet.”
Exercise is the single most powerful intervention. HIIT increases mitochondrial density by 40-80% in 8-12 weeks—an effect no supplement can match.
“More mitochondria always equals more energy.”
Dysfunctional mitochondria produce less ATP while generating more reactive oxygen species (ROS). Mitophagy (clearing damaged cells) is as vital as creating new ones.
“Mitochondrial dysfunction only affects the elderly.”
Sedentary lifestyles, processed foods, and chronic stress are causing dysfunction in young adults. Many 25-year-olds have worse function than active 55-year-olds.
“Cardio destroys mitochondria; only lift weights.”
Resistance training enhances volume and insulin sensitivity. Cardio increases density and oxidative capacity. The optimal approach combines both.
“High-dose antioxidants protect mitochondria.”
Moderate ROS production during exercise is the signal for biogenesis. Mega-dosing antioxidants (Vit C/E) peri-workout can reduce adaptations by 30-50%.
“Keto is always optimal for mitochondria.”
While keto enhances fat oxidation, it isn’t universally optimal. High-intensity performance often suffers, and some lose metabolic flexibility.
“Mitochondrial dysfunction is irreversible.”
Mitochondria are remarkably responsive. Studies show 60-80 year olds can increase density by 30-50% with just 12 weeks of proper training.
“You need expensive biohacks to optimize.”
The highest-leverage interventions are free: HIIT, cold showers, sunlight, and fasting. Expensive treatments (NAD+ IVs) offer marginal gains over these basics.
“Mitochondria are separate from general health.”
Mitochondria integrate energy, inflammation, and aging. Improving them simultaneously improves metabolic health, cognition, and cardiovascular function.
Why Mitochondria Determine Your Energy, Performance & Lifespan
Fatigue isn’t primarily a brain problem or a hormone problem. It is a cellular bioenergetics problem.
You experience it every afternoon: that inexplicable energy crash despite adequate sleep and nutrition. Or the inability to sustain focus for more than 90 minutes before needing stimulants. Or the training session that should feel manageable but leaves you completely depleted.
These aren’t character flaws or simply “getting older.” They are manifestations of mitochondrial dysfunction—impaired cellular energy production at the most fundamental level of human physiology.
Every cell in your body contains anywhere from dozens to thousands of mitochondria (cardiac muscle cells contain up to 5,000), and these organelles produce approximately 90% of cellular energy in the form of ATP (adenosine triphosphate).
- You produce ATP efficiently and sustain energy for hours without crashes.
- You recover quickly from physical and cognitive demands.
- You maintain metabolic flexibility (can utilize fats or glucose).
- You resist oxidative stress and age more slowly.
- You experience chronic fatigue despite adequate rest.
- You require constant stimulants (caffeine, sugar) to function.
- You crash after meals (glucose disposal impaired) and struggle with exercise recovery.
- You accumulate oxidative damage and accelerate biological aging.
The Mitochondrial Energy Crisis
Consider the sheer scale of ATP production: Your body produces and cycles through approximately your body weight in ATP every single day.
This massive energy demand explains why even modest dysfunction creates profound symptoms. A 20-30% reduction in mitochondrial ATP production creates persistent fatigue, impaired exercise capacity, and cognitive dysfunction.
The Salk Institute Connection: Research demonstrates that mitochondrial function predicts healthspan and lifespan more accurately than almost any other biomarker. Optimal function at age 60 yields performance matching sedentary 30-year-olds. Conversely, dysfunction at 30 predicts accelerated aging.
Mitochondria evolved approximately 1.5 billion years ago when an ancient bacterium was engulfed by a eukaryotic cell in an endosymbiotic event. This bacterial origin explains why mitochondria have their own DNA (circular, like bacteria), why they replicate independently, and why they are so sensitive to environmental stressors.
For millions of years, function was optimized by evolutionary pressures: Regular physical activity, frequent fasting, nutrient-dense whole foods, and circadian alignment. Modern life systematically undermines every factor that maintained this health.
The 6 Modern Villains
Why modern life destroys mitochondrial function.
Lack of physical activity eliminates the primary signal for biogenesis. Without energy demand, the body has no reason to maintain density. Desk workers typically have 40-60% lower mitochondrial density in skeletal muscle.
Industrial processing removes critical cofactors (B vitamins, magnesium, CoQ10) while adding damaging compounds like oxidized seed oils. The average diet provides less than 50% of optimal mitochondrial nutrients.
Sustained cortisol elevation impairs function by reducing biogenesis, increasing oxidative stress, and disrupting calcium regulation. Mitochondria interpret chronic stress as “emergency mode,” sacrificing long-term maintenance.
Mitochondrial repair and regeneration occur primarily during deep sleep. Chronic sleep restriction reduces density by 20-30%. Each hour of sleep debt creates measurable mitochondrial dysfunction.
Heavy metals, pesticides, and BPA directly damage mitochondrial DNA, which lacks the histone protection of nuclear DNA. They are particularly vulnerable to accumulated toxic burden.
Circadian misalignment from blue light exposure disrupts mitochondrial function by impairing master clock genes. Mitochondria have their own circadian rhythms that must coordinate with cellular clocks.
“This perfect storm of modern stressors explains epidemic levels of fatigue. But the extraordinary plasticity of mitochondria means interventions can rapidly restore function.”
Mitochondrial health is the foundation of human energy and longevity. Every symptom of “low energy” ultimately traces to Impaired Cellular ATP.
The ATP Energy Currency System
ATP (adenosine triphosphate) is the universal energy currency of all living cells. Understanding its production is fundamental to optimizing mitochondrial health and sustaining peak performance.
ATP consists of Adenine, Ribose, and Three Phosphate Groups connected by high-energy bonds. When the terminal bond breaks (ATP → ADP + Pi), approximately 7.3 kcal/mol of energy is released.
Biological Drivers
- Muscle contraction
- Protein synthesis
- Active membrane transport
Systemic Signaling
- DNA/RNA synthesis
- Neurotransmitter release
- Cellular signaling
The Three ATP Production Systems
Operates for 0-10 seconds of maximum effort. Powered by stored ATP and creatine phosphate. Recovery requires 3-5 minutes. No mitochondrial involvement.
Dominant for 10 seconds to 3 minutes. Yields 2 ATP per glucose anaerobically, producing lactate as a byproduct. Requires 30-90 minutes for full clearance.
Provides 90-95% of ATP during rest. Yields 36-38 ATP per glucose and 106-129 ATP per fatty acid. Complete mitochondrial dependence; this is the primary target for metabolic optimization.
The Molecular Machinery
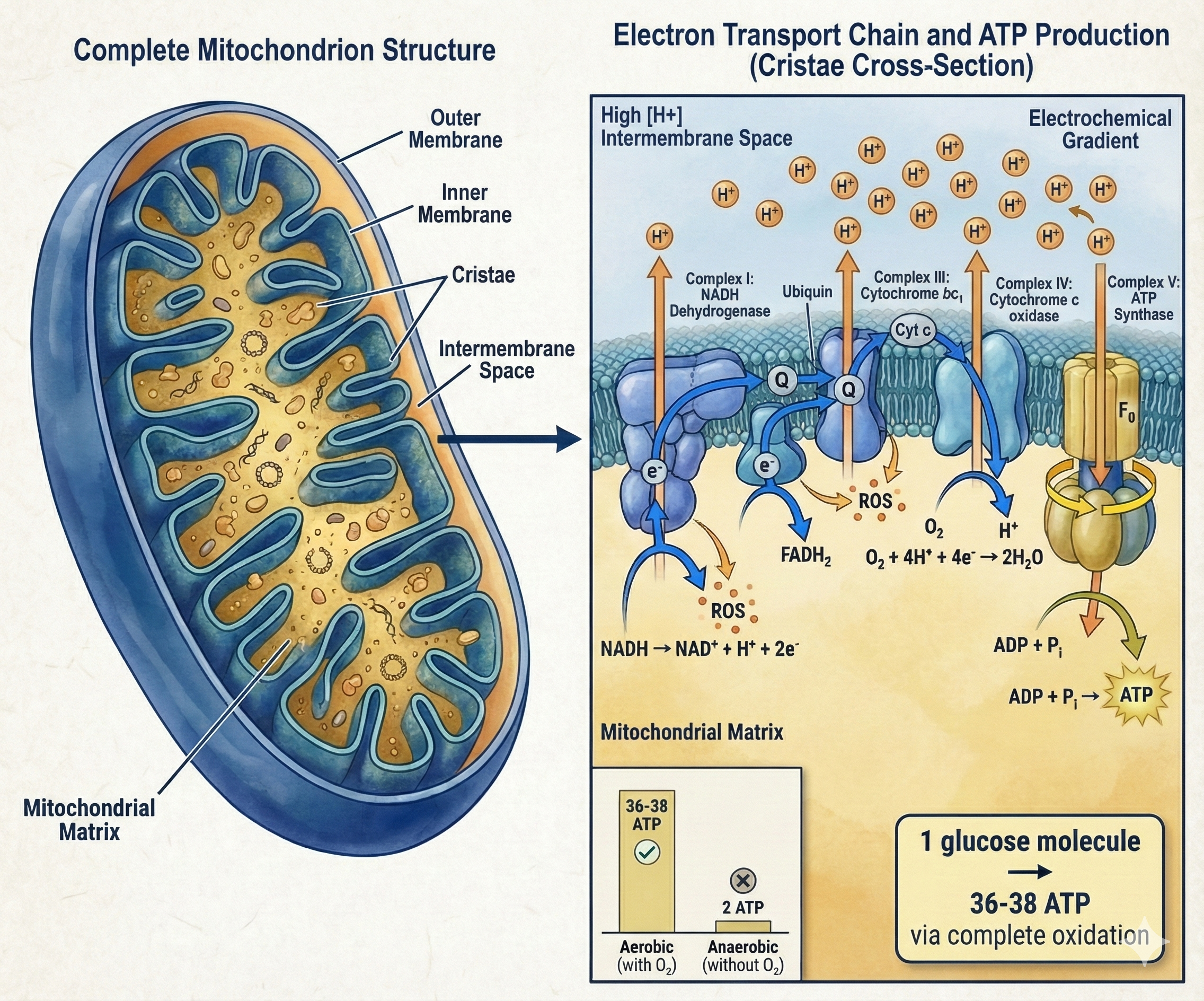
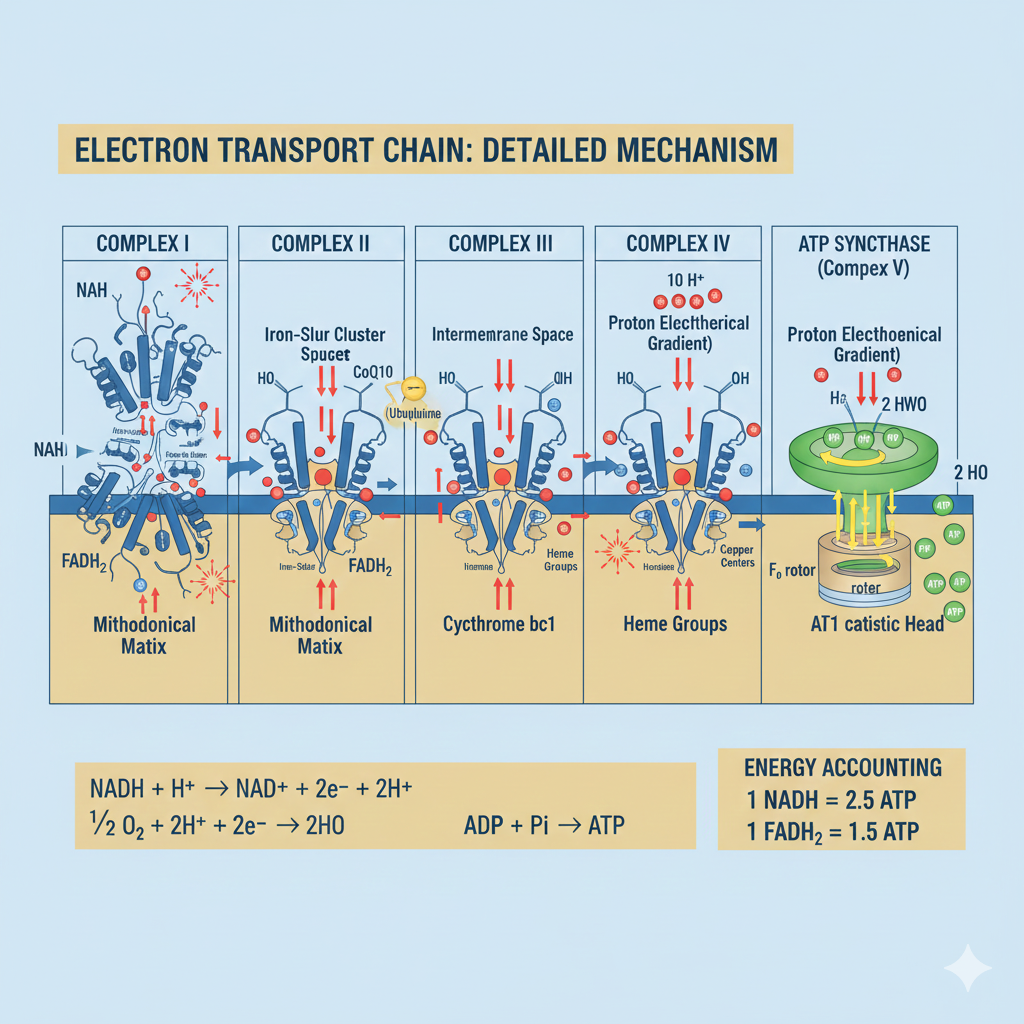
The Electron Transport Chain (ETC) is a series of protein complexes embedded in the inner mitochondrial membrane that generates the majority of cellular energy through oxidative phosphorylation.
- Step 1: Fuel Oxidation — Carbohydrates and Fats are broken down into Acetyl-CoA, entering the TCA Cycle to produce high-energy electron carriers (NADH and FADH2).
- Step 2: Electron Transport — Electrons flow through Complex I–IV. CoQ10 and Cytochrome C act as mobile carriers. Oxygen serves as the final electron acceptor.
- Step 3: Proton Gradient — As electrons flow, protons (H+) are pumped to the intermembrane space, creating a proton-motive force of ~180-220 mV.
- Step 4: ATP Synthesis — Protons flow back through Complex V (ATP Synthase), a molecular rotor spinning at ~150 RPS to catalyze the creation of ATP.
Actual energy yield is approximately 30-32 ATP per glucose (accounting for proton leak). This represents a 40% capture efficiency—significantly higher than a standard combustion engine (~25%).
The Electron Transport Chain is exquisitely efficient machinery. You cannot bypass this system—you can only Optimize It.
Mitochondrial Biogenesis: Creating Power
Mitochondrial biogenesis is the process by which cells increase their energy production capacity. This master regulatory system is the primary mechanism through which exercise and fasting transform your physiology.
The Master Regulator: PGC-1α
PGC-1α is the master regulator that coordinates the expression of hundreds of genes required for new mitochondrial formation. It enhances fat oxidation, increases antioxidant enzymes (SOD2, catalase), and promotes angiogenesis.
- AMPK Activation: Triggered by energy depletion (high AMP/ATP ratio).
- Calcium Signaling: Triggered by muscle contraction (CaMKII pathway).
- Sirtuin (SIRT1) Activity: Triggered by high NAD+/NADH ratios.
The Four Primary Stimuli
HIIT provides the strongest stimulus, yielding 40–80% density increases in 8–12 weeks via maximal ATP depletion. Moderate Training provides a volume-dependent adaptation, requiring more time for similar results.
Fasting triggers a low-energy state that activates AMPK and SIRT1. Beyond biogenesis, this initiates critical quality control through mitophagy, clearing out “zombie” mitochondria.
Cold stress activates Brown Adipose Tissue (BAT), which possesses the highest mitochondrial density. It induces UCP1, dissipating the proton gradient as heat and improving systemic insulin sensitivity.
Heat stress upregulates Heat Shock Proteins (HSPs) to protect mitochondrial structures, while polyphenols like resveratrol and curcumin provide the mild pro-oxidant stimulus needed to trigger PGC-1α.
The Transcriptional Cascade
- Hours: Acute increase in PGC-1α expression.
- Days: Increased transcription of nuclear mitochondrial genes.
- Weeks: Measurable increase in ETC proteins and mtDNA replication.
- Months: 30–80% increase in total mitochondrial density.

Healthy cells maintain a dynamic balance between Fission (division for quality control) and Fusion (joining for energy efficiency). Consistent training ensures this cycle remains robust.
Mitochondrial biogenesis is not passive—it requires deliberate signaling through Energy Stress.
Mitophagy: The Ruthless Quality Control
Creating new mitochondria is only half the equation. High-functioning biology requires the selective removal of damaged power plants before they poison the cell with oxidative stress.
The PINK1/Parkin Pathway
- 1. Damage Detection: Dysfunctional mitochondria lose membrane potential (~180 mV), causing PINK1 to accumulate on the outer membrane.
- 2. Parkin Recruitment: PINK1 phosphorylates ubiquitin, recruiting the Parkin enzyme from the cytoplasm to the damaged site.
- 3. Ubiquitination: Parkin creates a molecular “eat me” signal on the mitochondrial surface.
- 4. Autophagosome Formation: A double-membrane structure engulfs the mitochondrion, isolating it from the cell.
- 5. Lysosomal Degradation: The structure fuses with a lysosome, breaking the organelle down into recyclable constituent molecules.
Interventions for Enhanced Mitophagy
Duration matters. Mitophagy requires nutrient signaling to drop (low insulin/amino acids), typically peaking during 24–48 hour fasts or consistent 16:8 protocols.
- Urolithin A (500–1000mg): Gut metabolite that bypasses fasting to directly induce mitophagy.
- NAD+ Precursors (NMN/NR): Restores SIRT1 activity, the primary deacetylase regulating mitochondrial cleaning.
- Spermidine (1–10mg): Induces autophagy via epigenetic mechanisms and protects cardiovascular health.
The Critical Balance
Optimal health requires the synergy of Vigorous Biogenesis (building) and Robust Mitophagy (cleaning). One without the other leads to either a low-energy state or an accumulation of toxic “zombie” mitochondria.

Mitochondrial quantity without quality is worthless—you need High-Functioning mitochondria.
Essential Mitochondrial Cofactors
Mitochondria require specific nutrients as cofactors for ATP synthesis. Deficiencies in these elements create physical bottlenecks in energy production that no amount of training can override.
1. The B-Vitamin Complex
- B1 (Thiamine): Required for pyruvate dehydrogenase to convert pyruvate to Acetyl-CoA. Deficiency reduces carbohydrate-derived ATP by 40–60%. Dose: 1.5–100mg daily.
- B2 (Riboflavin): A structural component of FAD (electron carrier). Essential for Complex I and II function; deficiency stalls the entire ETC. Dose: 10–20mg daily.
- B3 (Niacin/NMN/NR): The precursor to NAD+. NAD+ levels decline 50% between age 20 and 80, directly impairing sirtuin-mediated repair and electron transport.
- B5 (Pantothenic Acid): A component of Coenzyme A. Without it, you cannot form Acetyl-CoA from any fuel source (fats, carbs, or proteins).
- B12 (Cobalamin): Required for odd-chain fatty acids to enter the TCA cycle. Deficiency causes toxic methylmalonic acid accumulation and energy failure. Dose: 500–1000mcg.
2. Critical Minerals
- Magnesium: The single most important mineral. Stabilizes the ATP-Mg complex (the active form of energy). Deficiency reduces ATP production by 30–40% and impairs utilization. Dose: 400–500mg (Glycinate or Malate).
- Iron (Ferritin): A structural component of the heme groups in Complexes II, III, and IV. If storage iron (Ferritin) is below 50 ng/mL, the mitochondria cannot effectively use oxygen.
- Selenium: A component of glutathione peroxidase. Protects mitochondria from the massive ROS production generated during oxidative phosphorylation. Dose: 100–200mcg.
3. Elite Energy Shuttles
Coenzyme Q10 (Ubiquinol): The primary electron shuttle. It moves electrons from Complexes I and II to Complex III. Supplementation (100–400mg) is critical after age 40 or when using statins, as endogenous production drops significantly.
L-Carnitine: The essential ferry for long-chain fatty acids. Without it, fats cannot cross the inner mitochondrial membrane for beta-oxidation, leading to metabolic inflexibility and fatigue.
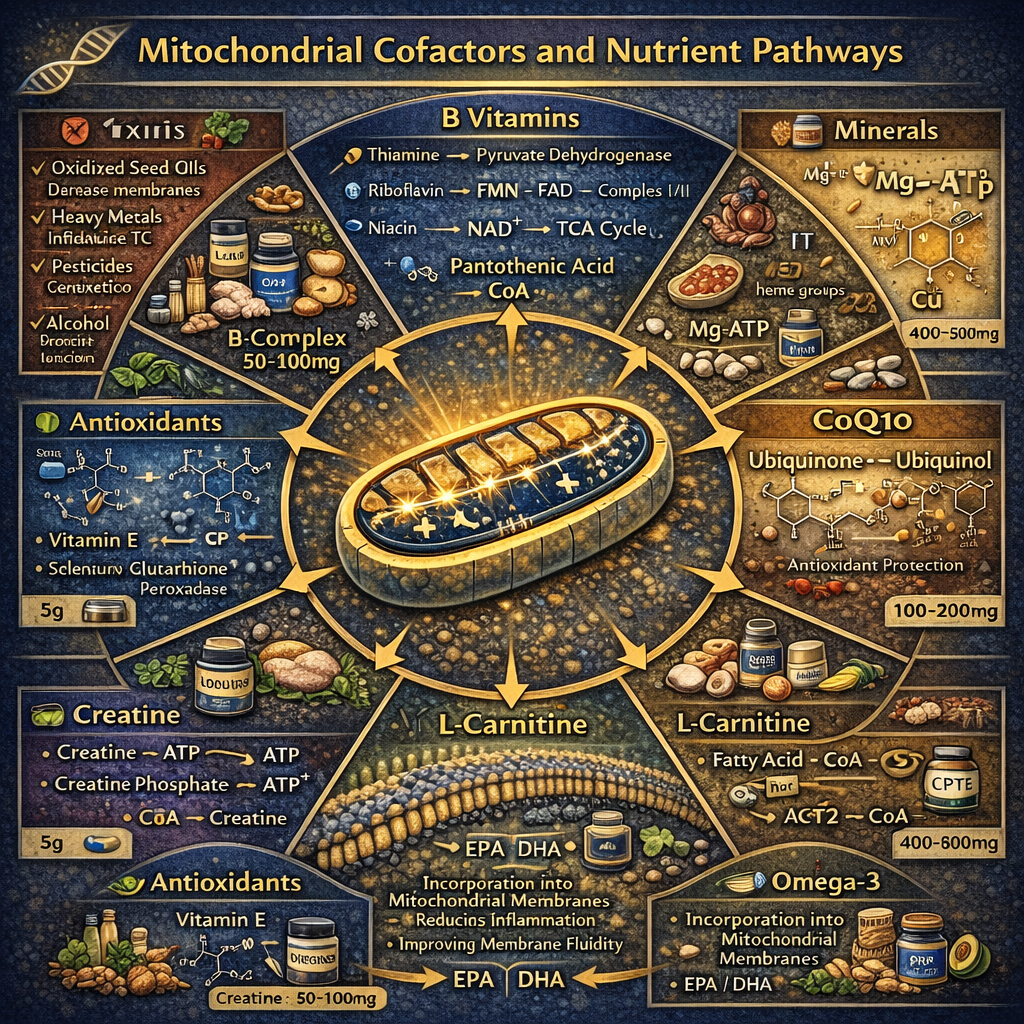
4. Membrane & Buffer Systems
- Omega-3 Fatty Acids (2–3g): Incorporates into the double membrane to improve fluidity and reduce lipid peroxidation. High Omega-6 seed oils do the opposite.
- Creatine Monohydrate (5g): Buffers the ATP/ADP ratio, providing instant regeneration during high-intensity output and reducing mitochondrial oxidative stress.
Optimization is impossible in the presence of active cellular damage. Avoid these “System Breakers”:
- Oxidized Seed Oils: (Soybean, Canola, Corn) These trigger lipid peroxidation in mitochondrial membranes.
- Excess Alcohol: Depletes NAD+ and directly damages mitochondrial DNA (mtDNA).
- Heavy Metals: Mercury, Lead, and Cadmium displace iron and zinc, essentially “clogging” the ETC complexes.
- Pharmaceutical Interference: Statins (reduce CoQ10 by 50%) and certain antibiotics (fluoroquinolones) which inhibit mitochondrial protein synthesis.
Mitochondrial nutrition isn’t about exotic supplements—it’s about providing the Fundamental Cofactors required for ATP synthesis.
Advanced Mitochondrial Optimization
Beyond the foundational pillars of nutrition and exercise lie advanced interventions designed to directly stimulate the Electron Transport Chain and bypass metabolic decay.
1. Photobiomodulation (PBM)
Using specific wavelengths (Red: 630–670nm; Near-Infrared: 810–850nm) to stimulate Cytochrome C Oxidase (Complex IV). Photon absorption at this rate-limiting step can increase ATP production by 150–200%.
- Pre-Exercise: Exposure improves power output by 5–8% and significantly accelerates recovery.
- Cognitive Focus: Transcranial NIR increases cerebral blood flow and memory processing speed.
- Protocol: 10–20 minutes per area, 5–6x weekly at a dose of 10–50 J/cm².
2. Hyperbaric Oxygen (HBOT)
By breathing 100% O2 at 1.5–3.0 atmospheres, you bypass hemoglobin saturation limits and increase dissolved oxygen in the plasma by 10–15x.
- Mitochondrial Impact: Oxygen is the final electron acceptor in the ETC. HBOT enhances ATP production in oxygen-limited tissues and paradoxically triggers biogenesis through hypoxia-inducible factors.
- Application: Typically 60–90 minute sessions. Effective for accelerated wound healing, injury recovery, and anti-aging maintenance.
3. Methylene Blue: The Electron Donor
At low doses (0.5–4mg/kg), Methylene Blue acts as an alternative electron carrier. It accepts electrons from NADH and donates them directly to Cytochrome C, effectively bypassing Complex I and III—the two primary sites of ROS production and energy failure.
Warning: Avoid if using SSRIs/MAOIs due to serotonin syndrome risk. Requires pharmaceutical-grade (USP) material.

4. NAD+ Restoration Therapies
- Oral Precursors (NMN/NR): Daily doses of 250–500mg increase NAD+ levels, restoring the fuel required for SIRT1 (mitophagy/biogenesis regulator) and DNA repair enzymes (PARPs).
- NAD+ IV Therapy: Direct infusion (250–1000mg) offers more rapid, dramatic effects than oral routes, though cost and discomfort are significant factors.
- CD38 Inhibition: Flavonoids like Apigenin (parsley/chamomile) and Quercetin inhibit the CD38 enzyme, which naturally consumes and depletes NAD+ as we age.
5. Emerging Compounds
- PQQ (10–20mg): A cofactor that may directly stimulate mitochondrial biogenesis beyond exercise signals.
- Alpha-Lipoic Acid (ALA): A dual-soluble antioxidant and cofactor for the pyruvate dehydrogenase complex.
- Urolithin A (500mg+): The most robust compound currently known for the direct induction of mitophagy in human skeletal muscle.
Advanced interventions provide incremental benefits, but the fundamentals provide 80% of the Results.
The Debiasing Mastery Protocol
A 90-day systematic programme to identify, counteract, and permanently reduce cognitive biases in your decision-making — from individual recognition through organisational transformation to permanent integration.
Based on Kahneman, Tetlock, Klein, and 40+ years of decision science research
Day Complete
Great work on your mitochondrial practice.
This is Elite-Level Optimization requiring significant time, financial, and recovery investment.
Part 5
Risks, Limitations
& The Dark Side
Where mitochondrial optimisation fails — and when pushing harder makes everything worse
Mitochondrial optimisation has a seductive logic: more energy means better performance. But biology doesn't reward brute-force approaches, and the gap between optimal stimulation and harmful overload is narrower than most people realise. Every intervention in this guide — from Zone 2 training to cold exposure to fasting protocols — has a therapeutic window. Below the threshold, you get nothing. Above it, you damage the very organelles you're trying to enhance.
What follows is an honest accounting of where mitochondrial protocols fail, who should avoid aggressive optimisation entirely, and the critical warning that separates evidence-based practice from the reckless experimentation that dominates biohacking culture.
Where Mitochondrial Optimisation Fails
These failure modes affect anyone pursuing mitochondrial optimisation. But for some, the risks are categorically different.
Who Should Not Optimise Aggressively
If any of these describe you, consult a medical professional before starting — not after symptoms appear.
Critical Warning
The "More Is Better" Trap in Mitochondrial Biohacking
The greatest danger in mitochondrial optimisation isn't any single protocol — it's the stacking mentality. Cold exposure plus fasting plus high-intensity training plus sauna plus sleep restriction to "maximise adaptation hours." Each intervention individually has evidence. Combined without recovery, they create a cumulative stress load that exceeds your body's adaptive capacity. This is the hormetic paradox: the dose that heals in isolation becomes the dose that harms in combination. Your mitochondria don't know you're "biohacking" — they just register unrelenting stress.
Self-Assessment — Check Any That Apply
- You stack multiple stressors (cold + fasting + HIIT) on the same day regularly
- Your HRV has been declining over the past month despite continued protocols
- You interpret fatigue as "not pushing hard enough" rather than a recovery signal
- You feel guilty taking rest days or reducing training intensity
Protection Against the Stacking Trap
- Never stack more than two hormetic stressors in a single day — cold OR fasting OR HIIT, not all three
- Track HRV trends weekly — declining baselines override any protocol schedule
- Include one full rest day per week with zero hormetic stress — this is when adaptation consolidates
- Monitor subjective markers: persistent fatigue, disrupted sleep, low libido, or mood changes are signals to deload immediately
Failure modes and exclusions describe individual risks. But the deepest limitations aren't personal — they're biological. This is Part 5 of the Mitochondrial Health blueprint.
The Limits of Lifestyle Mitochondrial Optimisation
Lifestyle interventions are powerful — but they operate within constraints that no amount of discipline can overcome. Understanding these ceilings prevents frustration and misdirected effort.
If you're experiencing persistent fatigue despite protocol adherence, these structural approaches address what individual optimisation cannot.
System-Level Solutions
- Comprehensive metabolic testing — organic acids, mitochondrial function panels, and nutrient status assessments identify specific bottlenecks that generic protocols miss
- Environmental audit — test your home and workplace for mould, VOCs, and heavy metal exposure; mitochondria are exquisitely sensitive to environmental toxins
- Periodised approach — cycle between building phases (high stimulus) and consolidation phases (reduced stress, emphasis on sleep and nutrition) rather than relentless optimisation
- Medical partnership — work with a functional medicine or sports medicine physician who can monitor biomarkers, adjust protocols based on objective data, and identify when symptoms indicate pathology rather than insufficient effort
- Psychological reframing — detach self-worth from optimisation metrics; the healthiest relationship with your biology involves acceptance of genetic constraints alongside strategic improvement within those boundaries
The goal was never maximum stress. It was optimal recovery — because adaptation happens in the rest.
The risks of mitochondrial optimisation are real: overtraining, supplement toxicity, fasting-induced hormonal disruption, cardiovascular danger from cold exposure, and the stacking mentality that treats your body as a machine rather than a biological system that requires recovery to adapt.
Your Questions Answered
16 research-backed answers covering energy science, performance optimization, and common myths — from understanding mitochondrial function to building your energy protocol.
No questions match your search
Try different keywords or
01What are mitochondria and why should I care about them?
Mitochondria are the microscopic power plants inside every cell that convert food into ATP — the universal energy currency your body uses for everything from thinking to moving — and your body produces approximately 65 kilograms of ATP every single day.
Every cell in your body (except red blood cells) contains hundreds to thousands of mitochondria. Neurons and muscle cells, which have the highest energy demands, contain the most. These organelles run a three-stage process: glycolysis (2 ATP in the cytoplasm), the Krebs cycle (2 ATP in the mitochondrial matrix), and the electron transport chain (34 ATP across the inner membrane). The critical insight is that 89% of your energy comes from the final stage — making electron transport chain efficiency the primary bottleneck for most people. When mitochondria are healthy and abundant, energy feels limitless. When they degrade — through age, sedentary living, poor nutrition, or chronic stress — fatigue sets in long before your muscles or mind should be tired. An estimated 85% of chronic disease is linked to mitochondrial dysfunction, making this arguably the most important and least understood component of performance optimisation.
Two surgeons perform identical 4-hour operations. One has high mitochondrial density from years of HIIT training and strategic nutrition — they finish alert and make sharp decisions throughout. The other, sedentary with degraded mitochondria, experiences progressive cognitive decline from hour 2 onward, relying on caffeine to mask the energy deficit. Same brain, same training, vastly different cellular power output.
Your mitochondria set the ceiling on every form of performance. Optimise them and the ceiling rises; neglect them and no amount of caffeine, motivation, or willpower compensates for the cellular energy deficit. **Citations:** 1. Wallace, D. C. (2005). A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer. *Genetics*, 171(4), 1751–1782. 2. Picard, M. et al. (2016). Mitochondrial functions modulate neuroendocrine, metabolic, inflammatory, and transcriptional responses to acute psychological stress. *PNAS*, 112(48), E6614–E6623.
02How does ATP production actually work?
ATP production runs through three sequential stages — glycolysis (fast but inefficient, producing 2 ATP), the Krebs cycle (2 ATP plus electron carriers), and the electron transport chain (34 ATP using oxygen) — with the final stage generating 89% of your total cellular energy.
Stage 1: Glycolysis occurs in the cytoplasm, splitting glucose into pyruvate and producing 2 ATP. This is fast and doesn't require oxygen — it's your emergency energy system and what powers brief high-intensity efforts. Stage 2: The Krebs cycle (citric acid cycle) runs in the mitochondrial matrix, processing acetyl-CoA from pyruvate or fatty acids, producing 2 ATP plus the electron carriers NADH and FADH₂. Stage 3: The electron transport chain (ETC) runs across the inner mitochondrial membrane, using those electron carriers plus oxygen to produce 34 ATP through oxidative phosphorylation. This is where the vast majority of energy comes from — and it's where most bottlenecks occur. When the ETC is inefficient, electrons "leak" and create reactive oxygen species (ROS) that damage the very mitochondria producing them. The total yield per glucose molecule is 38 ATP, but the system also runs on fatty acids (which produce even more ATP per molecule) and ketones.
Think of it like a three-stage rocket. Stage 1 (glycolysis) gets you off the ground quickly but burns out fast. Stage 2 (Krebs) extends the burn. Stage 3 (ETC) is the main engine that produces sustained power for orbit. Most people's "energy problem" is a Stage 3 efficiency problem — they have the fuel but their electron transport chain leaks energy as heat and oxidative damage rather than producing clean ATP.
Sustainable energy depends on Stage 3 efficiency. Everything in the protocol — HIIT, CoQ10, sleep, reducing oxidative damage — targets improving electron transport chain function. **Citations:** 1. Rich, P. R. (2003). The molecular machinery of Keilin's respiratory chain. *Biochemical Society Transactions*, 31(6), 1095–1105. 2. Chance, B. & Williams, G. R. (1955). Respiratory enzymes in oxidative phosphorylation. *Journal of Biological Chemistry*, 217(1), 383–393.
03What is substrate flexibility and why does it matter?
Substrate flexibility is your mitochondria's ability to switch seamlessly between glucose, fatty acids, and ketones as fuel sources based on availability and demand — and metabolic rigidity (being locked on glucose) is what creates the energy crashes that caffeine masks.
A metabolically flexible person fasting through a morning meeting runs smoothly on fatty acid oxidation and ketones. A metabolically rigid person in the same meeting experiences progressive brain fog as blood glucose drops because their mitochondria cannot efficiently switch fuel sources. The crashes most people attribute to "needing food" are actually substrate inflexibility — their mitochondria are locked on glucose and cannot transition to fat oxidation when glucose supply fluctuates. Building flexibility requires training both pathways: aerobic exercise (especially fasted Zone 2 cardio) forces fat oxidation adaptation, while occasional fasting extends the metabolic window. The result is mitochondria that produce stable energy regardless of when you last ate, eliminating the blood sugar roller coaster that drives caffeine dependency and afternoon crashes.
An executive who practises intermittent fasting and does fasted morning walks has built substrate flexibility. During a 6-hour negotiation with no meal break, their energy and cognition remain stable — mitochondria seamlessly shift from glucose to fatty acid oxidation. Their colleague, who eats every 3 hours, experiences progressive cognitive degradation from hour 3 onward as blood sugar falls and their rigid mitochondria have no alternative fuel pathway.
Train metabolic flexibility through fasted exercise and time-restricted eating. The goal is mitochondria that produce clean energy on any fuel available — not a brain that crashes when you miss lunch. **Citations:** 1. Goodpaster, B. H. & Sparks, L. M. (2017). Metabolic flexibility in health and disease. *Cell Metabolism*, 25(5), 1027–1036. 2. Muoio, D. M. (2014). Metabolic inflexibility: when mitochondrial indecision leads to metabolic gridlock. *Cell*, 159(6), 1253–1262.
04What is mitophagy and why is quality control so important?
Mitophagy is the cellular quality control process that identifies and recycles damaged mitochondria — and it's as important as creating new ones, because dysfunctional mitochondria don't just produce less ATP, they actively generate reactive oxygen species that destroy their healthy neighbours.
A single damaged mitochondrion with a leaky electron transport chain generates excessive ROS — reactive oxygen species that damage DNA, proteins, and lipid membranes in surrounding cells. Without mitophagy, these toxic units accumulate, creating an expanding zone of oxidative damage. It's analogous to a factory floor: one malfunctioning machine doesn't just reduce output — it throws sparks that damage nearby equipment. Mitophagy is triggered by fasting (activating AMPK and autophagy pathways), exercise (metabolic stress signals damaged units for removal), and sleep (major repair window). The combination of mitophagy (clearing damaged units) and biogenesis (creating new ones) through exercise produces a net population of high-quality mitochondria. This is why "more mitochondria" isn't automatically better — a large number of dysfunctional mitochondria is worse than a smaller number of highly functional ones.
A 16:8 intermittent fasting protocol activates autophagy/mitophagy pathways during the 16-hour fast. Combining this with morning HIIT (biogenesis trigger via PGC-1α) creates a dual signal: clear the damaged units overnight, build new ones in the morning. Over 12 weeks, this produces a mitochondrial population that's both denser and more efficient — explaining why people report both more energy and better recovery.
You need both creation (biogenesis) and destruction (mitophagy) for optimal mitochondrial health. Fasting clears the wreckage; exercise builds the replacements. Neither alone is sufficient. **Citations:** 1. Palikaras, K. et al. (2018). Mechanisms of mitophagy in cellular homeostasis, physiology and pathology. *Nature Cell Biology*, 20(9), 1013–1022. 2. Youle, R. J. & Narendra, D. P. (2011). Mechanisms of mitophagy. *Nature Reviews Molecular Cell Biology*, 12(1), 9–14.
05Why does mitochondrial function decline with age?
Mitochondrial density decreases approximately 10% per decade after 30 — but this decline is driven primarily by reduced physical activity, accumulated oxidative damage, and declining quality control rather than aging itself, and 60–80 year olds can increase density by 30–50% with just 12 weeks of proper training.
The "aging" of mitochondria follows a predictable cascade: reduced physical activity decreases the biogenesis signal (less PGC-1α activation), accumulated ROS damage impairs ETC efficiency (electron leak increases), declining NAD+ levels reduce repair capacity (sirtuins need NAD+ to function), and reduced mitophagy allows damaged units to accumulate. Each factor accelerates the others, creating an apparent inevitable decline that is actually largely behavioural. The landmark evidence: studies on elderly populations beginning structured exercise programmes show 30–50% increases in mitochondrial density within 12 weeks — demonstrating that the machinery for biogenesis remains intact even in advanced age. The decline isn't destiny; it's primarily a consequence of declining activity levels that remove the signal for mitochondrial maintenance.
A sedentary 65-year-old retiree begins a programme of 3x/week HIIT cycling and 2x/week resistance training. Muscle biopsy at baseline shows mitochondrial density comparable to a typical 80-year-old. After 16 weeks, density has increased 42% and is now comparable to a moderately active 45-year-old. The intervention reversed 20 years of "aging" in 4 months — because the decline was activity-driven, not age-driven.
Mitochondrial decline is modifiable at any age. The training response is preserved into your 80s. The earlier you start, the more you compound — but it's genuinely never too late. **Citations:** 1. Melov, S. et al. (2007). Resistance exercise reverses aging in human skeletal muscle. *PLoS ONE*, 2(5), e465. 2. Conley, K. E. et al. (2000). Oxidative capacity and ageing in human muscle. *Journal of Physiology*, 526(1), 203–210.
06What type of exercise most effectively builds new mitochondria?
High-intensity interval training (HIIT) is the most powerful biogenesis trigger — increasing mitochondrial density by 40–80% in 8–12 weeks by activating PGC-1α, the master regulator of mitochondrial creation.
PGC-1α (peroxisome proliferator-activated receptor gamma coactivator 1-alpha) is activated by the metabolic stress of intense exercise — specifically the energy demand exceeding current supply. HIIT creates this signal most efficiently: 30-second maximum efforts followed by 90-second recovery, repeated 6–8 rounds, 2–3 times per week. Total training time is approximately 20 minutes. Zone 2 aerobic training (60–70% max heart rate for 30–45 minutes) provides a complementary stimulus: it preferentially improves fat oxidation capacity and increases mitochondrial efficiency rather than raw density. The optimal programme combines both: HIIT 2–3x/week for density (building new power plants) and Zone 2 cardio 2–3x/week for efficiency (making existing power plants run cleaner). Resistance training adds volume improvements and insulin sensitivity. No supplement can replicate the biogenesis signal of exercise.
A 40-year-old does 20 minutes of HIIT cycling (8 × 30s sprints) on Monday and Thursday, plus 40-minute Zone 2 runs on Tuesday and Saturday. After 12 weeks, a muscle biopsy shows 55% increase in mitochondrial density and a 30% improvement in maximal oxygen uptake. The same person taking every marketed "mitochondrial supplement" without exercise would see near-zero improvement.
HIIT for density, Zone 2 for efficiency, resistance training for volume. Exercise is the single most powerful mitochondrial intervention — everything else is supplementary. **Citations:** 1. Hood, D. A. (2009). Mechanisms of exercise-induced mitochondrial biogenesis in skeletal muscle. *Applied Physiology, Nutrition, and Metabolism*, 34(3), 465–472. 2. Robinson, M. M. et al. (2017). Enhanced protein translation underlies improved metabolic and physical adaptations to different exercise training modes in young and old humans. *Cell Metabolism*, 25(3), 581–592.
07How does cold exposure improve mitochondrial function?
Cold stress activates brown adipose tissue (which is packed with mitochondria) and triggers the UCP1 protein pathway that drives both thermogenesis and mitochondrial biogenesis — 3–5 minutes at 10–15°C, 3–5 times weekly.
Brown adipose tissue (BAT) is distinct from white fat — it exists specifically to generate heat and is densely packed with mitochondria containing a unique protein called UCP1 (uncoupling protein 1). When activated by cold, UCP1 "uncouples" the electron transport chain, converting energy into heat rather than ATP. This uncoupling is a powerful biogenesis signal — the cell responds to the energy "waste" by building more mitochondria to compensate. Cold exposure also triggers norepinephrine release (200–300% increase), which independently activates PGC-1α in muscle tissue. The combined effect: more mitochondria in both BAT and muscle, improved thermogenesis (you become more cold-tolerant), enhanced metabolic rate, and increased norepinephrine-driven alertness and focus. The protocol is simple: cold showers (30–90 seconds of cold at the end), cold water immersion (10–15°C for 3–5 minutes), or outdoor cold exposure.
A knowledge worker adds 2 minutes of cold shower ending to their morning routine, 5 days per week. After 4 weeks, they notice improved morning alertness (norepinephrine effect), better cold tolerance (BAT activation), and sustained afternoon energy that previously required a third coffee. The mitochondrial biogenesis effect compounds over months, progressively raising their energy ceiling.
Cold is a free, powerful hormetic stressor. Start with 30 seconds of cold at the end of your shower and progress to 2–3 minutes. The discomfort is brief; the mitochondrial adaptation is lasting. **Citations:** 1. van Marken Lichtenbelt, W. D. et al. (2009). Cold-activated brown adipose tissue in healthy men. *New England Journal of Medicine*, 360(15), 1500–1508. 2. Šrámek, P. et al. (2000). Human physiological responses to immersion into water of different temperatures. *European Journal of Applied Physiology*, 81(5), 436–442.
08Can fasting really improve my energy?
Counterintuitively, yes — intermittent fasting (16:8) activates AMPK (the cellular energy sensor) and triggers mitophagy (clearing damaged mitochondria), resulting in a net population of more efficient power plants that produce cleaner energy with fewer crashes.
The paradox: temporarily reducing energy input improves energy output. When you fast, falling glucose and insulin activate AMPK, which signals the cell that energy is scarce. This triggers two responses: mitophagy (recycling damaged mitochondria for parts) and biogenesis (building new, efficient ones). Simultaneously, the body shifts toward fatty acid oxidation and ketone production — training substrate flexibility. Growth hormone surges 300–500% during fasting, supporting tissue repair without muscle catabolism. The key distinction is intermittent fasting (12–18 hours, regularly) versus prolonged fasting (24+ hours), which can become stressful and catabolic. For most people, a 16:8 pattern (eating window 12pm–8pm, fasting 8pm–12pm) provides the mitophagy and flexibility benefits without hormonal suppression. The critical requirement: adequate total caloric and nutrient intake within the feeding window. Fasting is a timing strategy, not a caloric restriction strategy.
An engineer switches from three meals plus snacks (7am–10pm eating window) to two substantial meals (12pm and 7pm, 16:8 fasting). Week 1 is uncomfortable as mitochondria adapt to fat oxidation. By week 3, morning mental clarity without food is noticeably superior to his previous caffeinated breakfast routine. At week 8, afternoon energy crashes have completely disappeared — his mitochondria have become substrate-flexible.
Start with 12-hour overnight fasts and gradually extend to 16:8. The initial discomfort is your mitochondria learning to use fat as fuel. Once adapted, energy becomes more stable than any meal-dependent schedule. **Citations:** 1. Mattson, M. P. et al. (2017). Impact of intermittent fasting on health and disease processes. *Ageing Research Reviews*, 39, 46–58. 2. Ho, K. Y. et al. (1988). Fasting enhances growth hormone secretion and amplifies the complex rhythms of growth hormone secretion in man. *Journal of Clinical Investigation*, 81(4), 968–975.
09What does morning sunlight have to do with mitochondrial function?
Mitochondria have their own circadian rhythms tied to light exposure — morning sunlight primes cytochrome c oxidase in the electron transport chain for peak daytime ATP output, and misaligned light signals reduce energy production independent of sleep quality.
Cytochrome c oxidase (Complex IV of the ETC) is a chromophore — it directly absorbs specific wavelengths of light, particularly red and near-infrared. Morning sunlight contains these wavelengths alongside the blue light that sets your circadian clock. When you get outdoor light exposure in the morning, you're simultaneously anchoring your circadian rhythm (via the suprachiasmatic nucleus) and priming your mitochondrial machinery for peak performance (via direct photon absorption in Complex IV). This is why red/near-infrared light therapy (photobiomodulation) at 660nm and 850nm can boost ATP production by 150–200% in targeted tissues. But the free version — morning sunlight — provides the full spectrum. Conversely, artificial indoor lighting throughout the day with no outdoor exposure leaves mitochondrial circadian signalling misaligned, contributing to the pervasive low-grade fatigue of modern indoor work.
A programmer who works from home starts taking a 15-minute morning walk in natural light before beginning work. Within two weeks, they report "an energy upgrade that feels different from caffeine — more like my brain is actually running faster, not just less tired." The photobiomodulation effect on their mitochondrial ETC is compounding with the circadian alignment benefit.
10–20 minutes of morning outdoor light is simultaneously your circadian anchor and your mitochondrial priming signal. No light therapy device fully replicates what sunlight provides for free. **Citations:** 1. Hamblin, M. R. (2016). Photobiomodulation or low-level laser therapy. *Journal of Biophotonics*, 9(11–12), 1122–1124. 2. Karu, T. I. (2008). Mitochondrial signaling in mammalian cells activated by red and near-IR radiation. *Photochemistry and Photobiology*, 84(5), 1091–1099.
10Can high-dose antioxidants actually harm my mitochondria?
Yes — mega-dosing antioxidants like vitamin C and vitamin E around exercise can blunt the very ROS signalling that triggers mitochondrial adaptation, reducing the benefits of training by 30–50%.
This is one of the most counterintuitive findings in exercise physiology. Moderate reactive oxygen species production during exercise is not merely damage — it's the adaptation signal. ROS activate transcription factors (including PGC-1α and Nrf2) that drive mitochondrial biogenesis, antioxidant enzyme upregulation, and cellular stress resistance. When you flood the system with exogenous antioxidants before or after exercise, you quench this signal before the cell can respond to it. Multiple studies have shown that supplementing with 1,000mg vitamin C and 400IU vitamin E around exercise significantly reduces improvements in VO₂max, mitochondrial density, and insulin sensitivity. The correct approach: get antioxidants from whole foods (berries, leafy greens, nuts) which provide moderate, balanced levels, and allow your endogenous antioxidant systems (superoxide dismutase, glutathione peroxidase, catalase) to strengthen through the hormetic exercise stimulus.
A runner training for a marathon takes 1,000mg vitamin C and 400IU vitamin E daily "for recovery." After 12 weeks, their VO₂max improvement is only 60% of what matched controls who took no supplements achieved. The antioxidants didn't help recovery — they prevented adaptation by blunting the training signal.
Eat antioxidant-rich whole foods but skip mega-dose supplements, especially around exercise. Your body's own antioxidant systems need the hormetic signal to get stronger. Artificially quenching ROS is like turning off the fire alarm instead of fireproofing the building. **Citations:** 1. Ristow, M. et al. (2009). Antioxidants prevent health-promoting effects of physical exercise in humans. *PNAS*, 106(21), 8665–8670. 2. Gomez-Cabrera, M. C. et al. (2008). Oral administration of vitamin C decreases muscle mitochondrial biogenesis and hampers training-induced adaptations in endurance performance. *American Journal of Clinical Nutrition*, 87(1), 142–149.
11What supplements actually support mitochondrial function?
The evidence-backed mitochondrial stack includes CoQ10 (100–200mg ubiquinol as the essential ETC electron carrier), magnesium (400–500mg for ATP-Mg complex stability), creatine (5g for rapid ATP regeneration), and omega-3s (2–3g for membrane integrity) — everything else is secondary.
CoQ10 (ubiquinone/ubiquinol) is a mandatory electron carrier between Complexes I/II and III of the ETC. Natural production declines with age, making supplementation increasingly important after 40. Ubiquinol (reduced form) has superior bioavailability. Take with fats for absorption. Magnesium is required for ATP to be biologically active — ATP exists as an ATP-Mg complex in the body. Without adequate magnesium, you can produce ATP but it can't function. Glycinate and threonate forms have the best bioavailability for brain and muscle. Creatine monohydrate donates a phosphate group to ADP, regenerating ATP instantly without oxygen — essentially a rapid ATP buffer for high-intensity demands. It also supports brain energy during cognitive load. Omega-3 fatty acids (EPA/DHA) maintain the integrity of mitochondrial membranes, particularly cardiolipin, which is essential for ETC function. Beyond the core four, PQQ (pyrroloquinoline quinone) shows emerging evidence for biogenesis stimulation, and alpha-lipoic acid supports both mitochondrial function and antioxidant recycling.
A 50-year-old executive adds the core mitochondrial stack: 200mg ubiquinol, 400mg magnesium glycinate before bed, 5g creatine daily, and 3g fish oil. After 6 weeks (alongside existing exercise programme), sustained focus extends by approximately 90 minutes daily and afternoon energy crashes have resolved. Total cost: roughly £60/month for a measurable quality-of-life improvement.
CoQ10, magnesium, creatine, omega-3s — in that order of priority. These aren't exotic biohacks; they're essential cofactors that many people are deficient in. Fix the deficiencies before chasing exotic compounds. **Citations:** 1. Rosenfeldt, F. L. et al. (2007). Coenzyme Q10 in the treatment of hypertension: a meta-analysis of the clinical trials. *Journal of Human Hypertension*, 21(4), 297–306. 2. Avgerinos, K. I. et al. (2018). Effects of creatine supplementation on cognitive function of healthy individuals. *Experimental Gerontology*, 108, 166–173.
12Why are seed oils bad for mitochondrial health?
Industrially processed seed oils (soybean, corn, canola, safflower) are high in polyunsaturated omega-6 fatty acids that are uniquely vulnerable to lipid peroxidation — and when these oxidised fats integrate into mitochondrial membranes, they damage cardiolipin, the phospholipid that the electron transport chain depends on.
Cardiolipin is a unique phospholipid found almost exclusively in the inner mitochondrial membrane, where it anchors the ETC complexes in their optimal configuration. Cardiolipin's function depends on its fatty acid composition — healthy cardiolipin contains predominantly linoleic acid (an omega-6), but when this oxidises (which it does readily when dietary omega-6 intake is excessive), it disrupts ETC complex assembly and increases electron leak. The result: less ATP production and more ROS damage. Seed oils contribute to this through sheer quantity — they now comprise 10–20% of calories in Western diets, a dramatic increase from the 2–3% of pre-industrial diets. Replacing seed oils with more stable fats (olive oil, avocado oil, butter, coconut oil, animal fats) reduces the substrate for lipid peroxidation and protects mitochondrial membrane integrity. This doesn't mean all omega-6 is bad — it's the industrial processing and excessive quantity that creates the problem.
A family audits their kitchen and discovers seed oils in their cooking oil, salad dressing, mayonnaise, crackers, and restaurant meals — an estimated 15% of total calories from oxidation-prone omega-6. Switching to olive oil and butter for cooking, making dressings at home, and cooking more meals from scratch reduces omega-6 intake by approximately 60%. While impossible to isolate the mitochondrial effect in one family, the intervention aligns with the mechanisms that protect cardiolipin integrity.
Replace industrial seed oils with olive oil, avocado oil, butter, and coconut oil. This protects mitochondrial membrane integrity and reduces the oxidative burden on your cellular power plants. **Citations:** 1. Claypool, S. M. & Koehler, C. M. (2012). The complexity of cardiolipin in health and disease. *Trends in Biochemical Sciences*, 37(1), 32–41. 2. DiNicolantonio, J. J. & O'Keefe, J. H. (2018). Omega-6 vegetable oils as a driver of coronary heart disease: the oxidized linoleic acid hypothesis. *Open Heart*, 5(2), e000898.
13Is NAD+ supplementation worth the hype?
NAD+ is genuinely critical for mitochondrial function and declines 50% between ages 40 and 60 — but the most cost-effective way to restore it is through exercise, fasting, and adequate niacin intake, not expensive IV infusions or NMN supplements.
Nicotinamide adenine dinucleotide (NAD+) is an essential coenzyme in every stage of ATP production and a required substrate for sirtuins — the cellular repair enzymes that maintain mitochondrial quality and DNA integrity. NAD+ decline with age is well-documented and contributes to both mitochondrial dysfunction and accelerated aging. The supplement industry has responded with NMN (nicotinamide mononucleotide) and NR (nicotinamide riboside) as oral NAD+ precursors, plus direct NAD+ IV infusions at $500–1,000 per session. While these raise blood NAD+ levels, the evidence that this translates to meaningful clinical outcomes in humans (as opposed to mice) is still emerging. What reliably increases NAD+ for free: exercise (activates NAMPT, the rate-limiting enzyme in NAD+ synthesis), fasting (AMPK activation upregulates NAD+ production), and adequate dietary niacin/tryptophan. These also provide dozens of other benefits that supplements don't.
A biohacker spending £300/month on NMN supplements while remaining sedentary is addressing the NAD+ decline without fixing the lifestyle factors that caused it. Their active colleague who does HIIT 3x/week, fasts 16:8, and takes a basic niacin supplement (£5/month) likely has comparable or superior NAD+ levels — along with the exercise-driven benefits of mitochondrial biogenesis, improved insulin sensitivity, and cardiovascular health that no supplement provides.
NAD+ matters. But master the free interventions (exercise, fasting, niacin) before spending hundreds on supplemental precursors. The evidence base for NMN/NR in humans is promising but not yet definitive enough to justify premium pricing over foundational lifestyle optimisation. **Citations:** 1. Massudi, H. et al. (2012). Age-associated changes in oxidative stress and NAD+ metabolism in human tissue. *PLoS ONE*, 7(7), e42357. 2. Yoshino, J. et al. (2018). NAD+ intermediates: the biology and therapeutic potential of NMN and NR. *Cell Metabolism*, 27(3), 513–528.
14What foods are best for mitochondrial health?
Prioritise fatty fish (omega-3s for membrane integrity), organ meats (CoQ10, B-vitamins, iron), colourful vegetables (polyphenols that activate Nrf2 antioxidant pathways), and adequate protein (amino acids for mitochondrial protein synthesis) — while eliminating processed seed oils and refined sugar.
The mitochondrial nutrition framework targets four systems: membrane integrity (omega-3 fatty acids from wild salmon, sardines, mackerel — DHA directly incorporates into cardiolipin), ETC cofactors (CoQ10 from organ meats and beef, B-vitamins from eggs and leafy greens, iron from red meat and liver, magnesium from dark chocolate and pumpkin seeds), antioxidant signalling (polyphenols from berries, dark leafy greens, and cruciferous vegetables activate the Nrf2 pathway — the master regulator of endogenous antioxidant production), and fuel quality (stable fats from olive oil, avocado, butter, and coconut oil that resist peroxidation). Foods to minimise: processed seed oils (membrane damage), refined sugars (insulin spikes that impair mitochondrial function), and ultra-processed foods (combination of damaged fats, sugar, and chemical additives).
A practical daily template: eggs cooked in butter for breakfast (choline, B-vitamins, stable fat), wild salmon with roasted vegetables and olive oil for lunch (omega-3s, polyphenols, cofactors), and grass-fed steak with sweet potato and dark leafy greens for dinner (CoQ10, iron, magnesium, fibre). Total cost increase over a processed diet is modest; the mitochondrial support is transformative.
Eat real food, emphasise fatty fish and organ meats, cook with stable fats, and load colourful vegetables. This provides the membrane building blocks, ETC cofactors, and antioxidant signalling your mitochondria need — no supplement stack required. **Citations:** 1. Gómez-Pinilla, F. (2008). Brain foods: the effects of nutrients on brain function. *Nature Reviews Neuroscience*, 9(7), 568–578. 2. Shen, Q. & Pierce, J. D. (2015). Supplementation of coenzyme Q10 among patients with type 2 diabetes mellitus. *Healthcare*, 3(2), 296–309.
15What's the simplest way to start improving my mitochondrial health today?
Three free interventions, starting today: a 15-minute morning walk in sunlight (circadian priming + photobiomodulation), one HIIT session this week (20 minutes, 6–8 sprints — the most powerful biogenesis trigger), and end tomorrow's shower with 30 seconds of cold water (BAT activation + norepinephrine).
The beauty of mitochondrial optimisation is that the highest-leverage interventions are free. Morning sunlight primes your ETC for daytime output and sets your circadian rhythm. A single HIIT session activates PGC-1α and begins the biogenesis cascade within hours. Cold exposure activates brown fat mitochondria and triggers norepinephrine-driven alertness. Layer these over the first two weeks before adding anything else. Week 3: begin 12-hour overnight fasts (8pm–8am) to activate mitophagy. Week 4: add the foundational supplement stack (CoQ10, magnesium, creatine, omega-3). Week 5–6: extend fasts to 16:8 and add Zone 2 cardio. This progressive approach builds each adaptation on the last without overwhelming your system.
Week 1: Morning walk (15 min) + cold shower ending (30 sec) + one HIIT session. Total daily time investment: 20 minutes. By week 2, most people report noticeably improved morning energy and a reduction in afternoon coffee dependency. These early signals indicate the mitochondrial machinery is responding — and each subsequent layer compounds on this foundation.
Sunlight, HIIT, and cold — all free, all evidence-based, all achievable today. Build from there. The expensive biohacks are marginal gains on top of these fundamentals, not replacements for them. **Citations:** 1. Hood, D. A. (2009). Mechanisms of exercise-induced mitochondrial biogenesis in skeletal muscle. *Applied Physiology, Nutrition, and Metabolism*, 34(3), 465–472. 2. van Marken Lichtenbelt, W. D. et al. (2009). Cold-activated brown adipose tissue in healthy men. *New England Journal of Medicine*, 360(15), 1500–1508.
16How long before I notice improvements in my energy?
Norepinephrine-driven alertness from cold exposure and sunlight is immediate (day 1), substrate flexibility improvements emerge at weeks 2–3, and measurable mitochondrial density increases require 8–12 weeks of consistent training — with substantial restoration possible in 12–24 weeks even from a deeply depleted baseline.
The timeline reflects different biological mechanisms operating at different speeds. Neurotransmitter effects (norepinephrine from cold, serotonin from light) produce same-day improvements in alertness and mood. Metabolic adaptations — particularly the shift toward fat oxidation during fasting — take 2–4 weeks as enzymatic pathways upregulate. Mitochondrial biogenesis (physical creation of new mitochondria in response to exercise) begins within 24 hours of a HIIT session but requires 8–12 weeks of consistent stimulus for population-level density changes visible on muscle biopsy. ETC efficiency improvements (cleaner electron transfer, less ROS leak) compound over months as damaged mitochondria are cleared via mitophagy and replaced by new, well-functioning units. The subjective experience typically follows: week 1 — better mornings; week 3 — fewer afternoon crashes; week 8 — sustained all-day energy without caffeine dependency; week 12+ — a fundamentally different relationship with energy.
A 42-year-old consultant starts the protocol and tracks their daily energy on a 1–10 scale. Baseline average: 5.2. Week 2: 6.1 (morning improvement from sunlight/cold). Week 6: 7.0 (afternoon crashes reduced from fasting adaptation). Week 12: 8.1 (HIIT-driven biogenesis producing sustained output). Week 24: 8.7 (full system optimisation). The improvement is nonlinear — early gains are neurochemical, later gains are structural.
Expect immediate neurochemical benefits (days), metabolic flexibility gains (weeks), and structural mitochondrial improvements (months). The compound effect means week 12 feels like a completely different energy system than week 1. Patience and consistency are the only requirements. **Citations:** 1. Robinson, M. M. et al. (2017). Enhanced protein translation underlies improved metabolic and physical adaptations to different exercise training modes. *Cell Metabolism*, 25(3), 581–592. 2. Melov, S. et al. (2007). Resistance exercise reverses aging in human skeletal muscle. *PLoS ONE*, 2(5), e465.
You've explored all 16 questions
Ready to go deeper? The full Mitochondrial Health article provides comprehensive frameworks, implementation protocols, and advanced optimization systems.
Your Mitochondrial Transformation
You now possess a comprehensive, science-based framework for systematically optimizing your mitochondria—the cellular power plants determining your energy, performance, cognitive function, and longevity.
The Core Principles Revisited
- Foundation: Mitochondria are the source of human vitality. Every symptom of "low energy" traces back to impaired ATP production. You cannot hack around this fundamental biology.
- Exercise as Master Regulator: HIIT produces 40-80% increases in mitochondrial density. This molecular signaling from energy stress initiates the transcriptional cascade required for biogenesis.
- Quality Control: Optimization = Vigorous Biogenesis (Building) + Ruthless Mitophagy (Cleaning) + Cofactor Support (Nutrition).
- Recovery: Biogenesis occurs during recovery, not training. Sleep is the primary window for mitochondrial assembly and translation.
The Hierarchy of Impact
- The One Thing: Sleep 7-9 hours nightly with consistent timing.
- The Three Things: Add HIIT 2-3x weekly and eliminate industrial seed oils/toxins.
- Complete Optimization: Basic 30-Day Protocol → Advanced Protocol progression.
Measuring Success
Subjective: Waking refreshed, sustained energy for 12-16 hours, 4-6 hour deep work focus, and stable emotional mood.
Objective: VO2max gains (10-20%), strength improvements, reduced fasting glucose/HbA1c, and lower inflammatory markers.
Mistakes to Avoid
- Prioritizing supplements over lifestyle (sleep/training/nutrition).
- Ignoring recovery and HRV signals (Mitochondrial exhaustion).
- Pursuing extreme leanness that triggers metabolic adaptation.
- Obsessive perfectionism; 90% adherence with flexibility beats 100% with anxiety.
The Long Game: Compounding Benefits
Active 60-year-olds with optimized mitochondria have energy production capacity matching sedentary 30-year-olds. The decline is largely lifestyle-mediated.
- 30s: Prevent age-related decline; establish longevity habits.
- 40s-50s: Slow biological aging; preserve muscle and metabolic health.
- 60s+: Prevent frailty; maintain functional independence and healthspan.
This Week: Consistent sleep schedule + 3 training sessions + eliminate one toxin source.
This Month: Implement the full Basic 30-Day Protocol.
This Year: Establish optimization as a permanent lifestyle; retest performance metrics quarterly.
"Master your cellular energy production, and you master your biology. Master your biology, and you unlock your full human potential."
BEGIN TODAY.
“Fatigue makes cowards of us all.” — Vince Lombardi
What You Need to Remember
What actually determines how much energy you have — and where it goes.
Mitochondria set your ceiling
Every thought, every movement, every decision runs on mitochondrial ATP. Mitochondrial density and efficiency determine your sustainable energy output — everything else is downstream.
Explore: Module 1 — Mitochondrial Science →You run on three fuel systems
Phosphagen (10 seconds of explosive output), glycolytic (2 minutes of intense work), oxidative (hours of sustained effort). Mismatching the fuel system to the demand is why your energy feels inconsistent.
Explore: Module 1 — Energy System Map →Conversational-pace exercise builds power plants
Zone 2 training (60-70% max HR, you can still talk) is the most efficient stimulus for mitochondrial biogenesis — you're literally building new cellular power plants at a conversational pace.
Explore: Module 2 — Zone 2 Protocol →You're losing 20-30% to invisible drains
Poor sleep quality, chronic low-grade inflammation, subclinical nutrient gaps, and metabolic inflexibility leak 20-30% of your available energy without producing any obvious fatigue signal.
Explore: Module 3 — Energy Leaks →Can you run on both fuels?
Metabolic flexibility — efficiently switching between glucose and fat oxidation — determines your resistance to energy crashes, fasting tolerance, and sustained cognitive output. Most people are locked on glucose.
Explore: Module 3 — Metabolic Flexibility →Every stimulant is a loan with interest
Caffeine doesn't create energy. It blocks the tiredness signal. Sugar doesn't sustain energy. It spikes and crashes. Both advance future reserves into the present — and the interest compounds.
Explore: Module 4 — Stimulant Trap →Brief stress builds; chronic stress destroys
Cold exposure, sauna, and fasting trigger hormetic adaptation — more mitochondria, better efficiency. But the dose matters: 30-90 seconds of cold builds. Chronic unrelenting stress degrades.
Explore: Module 4 — Hormesis Protocols →Three compounds with actual evidence
CoQ10 (electron transport chain), creatine (phosphagen buffer), and NMN/NR (NAD+ precursors) have mechanistic evidence at the mitochondrial level — not miracle pills, but targeted substrate support.
Explore: Module 5 — Supplement Stack →Energy is rhythmic, not flat
Ultradian cycles (90-minute peaks), circadian cycles (daily), and infradian cycles (weekly/monthly) mean sustainable energy requires rhythmic output and recovery — not constant grinding.
Explore: Module 5 — Energy Periodization →Energy is the compound of everything else
Sleep + nutrition + movement + stress regulation + strategic recovery. There's no energy hack. There's an energy system, and every article in this pillar feeds into it.
Explore: Module 5 — Engineering Energy →Continue Your Journey
References
0 sources cited — journal articles, foundational texts, and landmark studies in mitochondrial biogenesis, oxidative phosphorylation, mitophagy, metabolic flexibility, and cellular energy optimization
- 1(2014). Mitochondrial form and function. Nature, 505(7483), 335–343.
- 2(2018). The multifaceted contributions of mitochondria to cellular metabolism. Nature Cell Biology, 20(7), 745–754.
- 3(2013). Bioenergetics 4. Academic Press. Book
- 4(1961). Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature, 191, 144–148.
- 5(2012). The oxidative phosphorylation system in mammalian mitochondria. Advances in Experimental Medicine and Biology, 942, 3–37.
- 6(2011). Metabolic control of mitochondrial biogenesis through the PGC-1 family regulatory network. Biochimica et Biophysica Acta, 1813(7), 1269–1278.
- 7(2003). Peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1 alpha): Transcriptional coactivator and metabolic regulator. Endocrine Reviews, 24(1), 78–90.
- 8(2002). Adaptations of skeletal muscle to exercise: Rapid increase in the transcriptional coactivator PGC-1. FASEB Journal, 16(14), 1879–1886.
- 9(2010). A practical model of low-volume high-intensity interval training induces mitochondrial biogenesis in human skeletal muscle. Journal of Physiology, 588(6), 1011–1022.
- 10(2007). Exercise-induced mitochondrial biogenesis begins before the increase in muscle PGC-1alpha expression. Journal of Biological Chemistry, 282(1), 194–199.
- 11(2008). Regulation by exercise of skeletal muscle content of mitochondria and GLUT4. Journal of Physiology and Pharmacology, 59(Suppl 7), 5–18.
- 12(2006). Coordination of metabolic plasticity in skeletal muscle. Journal of Experimental Biology, 209(12), 2265–2275.
- 13(2012). Physiological adaptations to low-volume, high-intensity interval training in health and disease. Journal of Physiology, 590(5), 1077–1084.
- 14(2008). Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans. Journal of Physiology, 586(1), 151–160.
- 15(2003). Regulation of mitochondrial biogenesis in muscle by endurance exercise. Sports Medicine, 33(11), 783–793.
- 16(2016). The effect of training intensity on VO2max in young healthy adults: A meta-regression and meta-analysis. International Journal of Exercise Science, 9(2), 230–247.
- 17(2005). Very intense exercise-training is extremely potent and time efficient: A reminder. Journal of Applied Physiology, 98(6), 1983–1984.
- 18(2011). Mechanisms of mitophagy. Nature Reviews Molecular Cell Biology, 12(1), 9–14.
- 19(2018). Mechanisms of mitophagy in cellular homeostasis, physiology and pathology. Nature Cell Biology, 20(9), 1013–1022.
- 20(2018). Mitophagy and quality control mechanisms in mitochondrial maintenance. Current Biology, 28(4), R170–R185.
- 21(2007). Selective degradation of mitochondria by mitophagy. Archives of Biochemistry and Biophysics, 462(2), 245–253.
- 22(2013). The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death & Differentiation, 20(1), 31–42.
- 23(2006). Calorie restriction induces mitochondrial biogenesis and bioenergetic efficiency. Proceedings of the National Academy of Sciences, 103(6), 1768–1773.
- 24(2014). Fasting: Molecular mechanisms and clinical applications. Cell Metabolism, 19(2), 181–192.
- 25(2018). Flipping the metabolic switch: Understanding and applying the health benefits of fasting. Obesity, 26(2), 254–268.
- 26(2019). Effects of intermittent fasting on health, aging, and disease. New England Journal of Medicine, 381(26), 2541–2551.
- 27(2011). The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers. International Journal of Obesity, 35(5), 714–727.
- 28(2006). Mitochondrial function and toxicity: Role of the B vitamin family on mitochondrial energy metabolism. Chemico-Biological Interactions, 163(1-2), 94–112.
- 29(2013). Magnesium in disease prevention and overall health. Advances in Nutrition, 4(3), 378S–383S.
- 30(2006). Update on the relationship between magnesium and exercise. Magnesium Research, 19(3), 180–189.
- 31(2014). Review on iron and its importance for human health. Journal of Research in Medical Sciences, 19(2), 164–174.
- 32(2000). Iron status and exercise. American Journal of Clinical Nutrition, 72(2 Suppl), 594S–597S.
- 33(2018). Coenzyme Q10 supplementation in aging and disease. Frontiers in Physiology, 9, 44.
- 34(2014). Clinical applications of coenzyme Q10. Frontiers in Bioscience, 19, 619–633.
- 35(2001). Biochemical functions of coenzyme Q10. Journal of the American College of Nutrition, 20(6), 591–598.
- 36(2015). Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochimica et Biophysica Acta, 1851(4), 469–484.
- 37(2002). Omega-3 fatty acids in inflammation and autoimmune diseases. Journal of the American College of Nutrition, 21(6), 495–505.
- 38(2017). International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. Journal of the International Society of Sports Nutrition, 14, 18.
- 39(2018). Effects of creatine supplementation on cognitive function of healthy individuals: A systematic review of randomized controlled trials. Experimental Gerontology, 108, 166–173.
- 40(2018). L-Carnitine supplementation in recovery after exercise. Nutrients, 10(3), 349.
- 41(2011). L-Carnitine: Metabolic functions and meaning in humans life. Current Drug Metabolism, 12(7), 667–678.
- 42(2009). Identification and importance of brown adipose tissue in adult humans. New England Journal of Medicine, 360(15), 1509–1517.
- 43(2013). Cold acclimation recruits human brown fat and increases nonshivering thermogenesis. Journal of Clinical Investigation, 123(8), 3395–3403.
- 44(2013). Recruited brown adipose tissue as an antiobesity agent in humans. Journal of Clinical Investigation, 123(8), 3404–3408.
- 45(2004). Brown adipose tissue: Function and physiological significance. Physiological Reviews, 84(1), 277–359.
- 46(2012). The nuts and bolts of low-level laser (light) therapy. Annals of Biomedical Engineering, 40(2), 516–533.
- 47(1999). Primary and secondary mechanisms of action of visible to near-IR radiation on cells. Journal of Photochemistry and Photobiology B, 49(1), 1–17.
- 48(2016). Photobiomodulation of human adipose-derived stem cells using 810nm and 980nm lasers operates via different mechanisms of action. Biochimica et Biophysica Acta, 1861(6), 441–449.
- 49(2016). Proposed mechanisms of photobiomodulation or low-level light therapy. IEEE Journal of Selected Topics in Quantum Electronics, 22(3), 7000417.
- 50(2015). Association between sauna bathing and fatal cardiovascular and all-cause mortality events. JAMA Internal Medicine, 175(4), 542–548.
- 51(1988). The heat-shock proteins. Annual Review of Genetics, 22, 631–677.
- 52(2018). Therapeutic potential of NAD-boosting molecules: The in vivo evidence. Cell Metabolism, 27(3), 529–547.
- 53(2018). NAD+ intermediates: The biology and therapeutic potential of NMN and NR. Cell Metabolism, 27(3), 513–528.
- 54(2015). NAD+ metabolism and the control of energy homeostasis: A balancing act between mitochondria and the nucleus. Cell Metabolism, 22(1), 31–53.
- 55(2015). NAD+ in aging, metabolism, and neurodegeneration. Science, 350(6265), 1208–1213.
- 56(2019). The mitophagy activator urolithin A is safe and induces a molecular signature of improved mitochondrial and cellular health in humans. Nature Metabolism, 1(6), 595–603.
- 57(2016). Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nature Medicine, 22(8), 879–888.
- 58(2022). Direct supplementation with urolithin A overcomes limitations of dietary exposure and gut microbiome variability in healthy adults. European Journal of Clinical Nutrition, 76(2), 297–308.
- 59(2013). The hallmarks of aging. Cell, 153(6), 1194–1217.
- 60(2016). The mitochondrial basis of aging. Molecular Cell, 61(5), 654–666.
- 61(2005). Decline in skeletal muscle mitochondrial function with aging. Proceedings of the National Academy of Sciences, 102(15), 5618–5623.
- 62(2010). The effect of aging on human skeletal muscle mitochondrial and intramyocellular lipid ultrastructure. Journals of Gerontology Series A, 65(2), 119–128.
- 63(2004). The epidemiology of mitochondrial disorders—past, present and future. Biochimica et Biophysica Acta, 1659(2-3), 115–120.
- 64(2016). Mitochondrial diseases. Nature Reviews Disease Primers, 2, 16080.
- 65(2013). Prevention, diagnosis, and treatment of the overtraining syndrome: Joint consensus statement of the European College of Sport Science. Medicine & Science in Sports & Exercise, 45(1), 186–205.
- 66(2021). The immune system and overtraining in athletes: Clinical implications. Acta Clinica Croatica, 59(Suppl 1), 18–26.
- 67(2017). Metabolic flexibility in health and disease. Cell Metabolism, 25(5), 1027–1036.
- 68(2018). Metabolic flexibility as an adaptation to energy resources and requirements in health and disease. Endocrine Reviews, 39(4), 489–517.
- 69(2008). Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production. Physiological Reviews, 88(4), 1243–1276.
- 70(2009). Antioxidants prevent health-promoting effects of physical exercise in humans. Proceedings of the National Academy of Sciences, 106(21), 8665–8670.
- 71(2003). Mitochondrial dysfunction in the elderly: Possible role in insulin resistance. Science, 300(5622), 1140–1142.
- 72(2012). Mitochondrial fission contributes to mitochondrial dysfunction and insulin resistance in skeletal muscle. Molecular and Cellular Biology, 32(2), 309–319.
- 73(2018). Response of mitochondrial fusion and fission protein gene expression to exercise in rat skeletal muscle. Biochimica et Biophysica Acta, 1800(3), 250–256.
- 74(2020). Sleep loss can cause death through accumulation of reactive oxygen species in the gut. Cell, 181(6), 1307–1328.
- 75(2012). Mitochondrial dysfunction in neurodegenerative diseases. Journal of Pharmacology and Experimental Therapeutics, 342(3), 619–630.
- 76(2011). Mitochondria: The next (neurode)generation. Neuron, 70(6), 1033–1053.